How about an incentive to share this post? (You will help other colleagues find this blog)ĭownload and enjoy this complete and colored periodic table for you to edit and enjoy. Need an editable periodic table to edit? Maybe add your school logo, work team or anything else to maker your paper look cool?Īlong with basic atom / element information (like Oxygen valence electrons and all the other atomic data), it also comes with color coded info about: State (Gas, Liquid or Solid at room temperature), Groups/series details and much more. How a small number of atoms can be joined and form completely different substances.Video Are you having trouble understanding the basics of atomic elements? This video will walk you through: Want to learn more details and data about Oxygen (O)? Check my Elements Comprehensive List. Small amounts are made in the laboratory by electrolysis of water or heating potassium chlorate (KClO3) with manganese dioxide (MnO2) catalyst.ĭiscoveryDiscovered By: Joseph Priestly, Carl Wilhelm Scheele SourcesObtained primarily from liquid air by fractional distillation. 
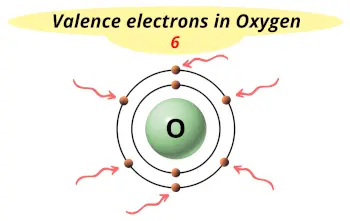
Name OriginGreek: oxys and genes, (acid former). Naturally occuring ozone (O3) in the upper atmosphere shields the earth from ultraviolet radiation. UsesUsed in steel making, welding, and supporting life. It is the most abundant element in the earth's crust, and makes up almost 21% of the atmosphere. Third most abundant element in the universe. In the case of Oxygen the valence electrons is 2. Now let's check the facts about Oxygen.ĭescriptionColorless, odorless, tasteless gas pale blue liquid. Ok but how many valence electrons does an atom of Oxygen have? A valence electron is an outer shell electron and may participate in the formation of a chemical bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
